Extractables and Leachables Testing Services Market to Reach USD 6341.16 Million by 2032: Maximize Market Research
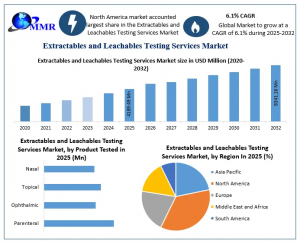
Extractables and Leachables Testing Services Market was valued at USD 4,189.49 million in 2025 and is projected to reach USD 6,341.16 million by 2032.
Maximize Market Research reveals explosive growth in E&L Testing Services as FDA, EMA regulations intensify!”
ROCKVILLE , MD, UNITED STATES, March 23, 2026 /EINPresswire.com/ -- Global Extractables and Leachables (E&L) Testing Services Market size was valued at USD 4189.49 Million in 2025 and is projected to expand at a CAGR of 6.1% from 2026 to 2032, reaching approximately USD 6341.16 Million by 2032.— Maximize Market Research
Get Full PDF Sample Copy of Report: (Including Full TOC, List of Tables & Figures, Chart) @ https://www.maximizemarketresearch.com/request-sample/82518/
Extractables & Leachables (E&L) Testing Services Market 2025: Driving Pharma Safety, Regulatory Compliance, and Analytical Innovation Worldwide
Global Extractables and Leachables (E&L) Testing Services Market Report 2025 provides an in-depth analysis of market trends, size, and forecasts through 2032. The industry is witnessing robust growth driven by stringent FDA, EMA, and ICH regulations and rising demand for SVP and LVP drug safety assessments. Adoption of advanced LC-MS/MS and GC-MS analytical platforms, multi-modal testing, AI-driven predictive toxicology, and innovative container-closure system evaluations is transforming the market landscape. Strategic collaborations between CROs and pharma companies, along with regional expansion in North America, Europe, and APAC, continue to shape the future of the global E&L Testing Services Market. Regulatory compliance, patient safety, and analytical innovation are key factors fueling market growth worldwide.
E&L Testing Services Market Driven by FDA/EMA Regulations and Rising SVP & LVP Drug Safety Demand
Extractables and Leachables (E&L) Testing Services Market is experiencing robust growth as stringent FDA, EMA, and ICH regulations compel pharmaceutical and biotech companies to adopt advanced analytical testing. Rising demand for small-volume parenteral (SVP) and large-volume parenteral (LVP) drug safety assessments, container-closure system analysis, and comprehensive E&L testing services highlights its critical role in patient safety, regulatory compliance, and toxicological risk mitigation.
E&L Testing Services Market Constrained by High Costs, Complex Methods, and Regional Standardization Challenges
Extractables and Leachables (E&L) Testing Services Market faces key challenges including high operational costs of advanced testing laboratories, complex analytical method development, and limited standardization across regions. Variability in drug formulations and container-closure materials increases testing complexity and turnaround time, restricting adoption among small and mid-sized pharmaceutical firms while emphasizing the urgent need for cost-effective, scalable E&L testing solutions globally.
E&L Testing Services Market Poised for Growth with APAC Expansion, AI Integration, and Personalized Medicine Demand
Extractables and Leachables (E&L) Testing Services Market is ripe with strategic growth opportunities. Rapid pharmaceutical manufacturing expansion in APAC, the rising adoption of personalized medicine and biologics, and the integration of AI-driven predictive toxicology models are driving demand. Additionally, strategic partnerships between pharma companies and specialized E&L testing labs enable customized container-closure system analysis, accelerated regulatory approvals, and enhanced drug safety assurance, positioning the market for global expansion and innovation leadership.
E&L Testing Services Market Trends: Early-Stage Testing, Green Packaging, QbD Integration, and Multi-Modal Solutions Driving Global Growth
Proactive Early-Stage E&L Testing Adoption Fuels Market Growth: The Extractables and Leachables (E&L) Testing Services Market is witnessing a shift toward preventive testing during drug development, enabling early detection of harmful leachables. This advanced container-closure system analysis reduces regulatory risks, enhances pharmaceutical safety compliance, and positions E&L testing as a critical tool for patient safety and quality assurance.
Sustainable and Low-Leachable Materials Drive Innovation: Increasing emphasis on green chemistry and eco-friendly packaging is reshaping the market. E&L testing services are now applied to evaluate low-leachable polymers, sustainable container-closure systems, and innovative drug packaging solutions, combining regulatory compliance with environmental responsibility, and creating new growth avenues for analytical testing labs globally.
Integration with Quality by Design (QbD) Accelerates Compliance and Efficiency: Leading pharmaceutical manufacturers are embedding E&L testing into QbD frameworks to anticipate chemical interactions in container-closure systems during early drug design. This proactive approach enhances drug safety assurance, regulatory compliance, and manufacturing efficiency, solidifying the role of state-of-the-art E&L testing services in modern pharmaceutical development.
Expansion of Multi-Modal, End-to-End E&L Testing Solutions: Testing providers are offering comprehensive, multi-modal services combining extractables, leachables, migration studies, and toxicological assessments. These bespoke E&L testing solutions cater to complex drug formulations, biologics, and innovative delivery systems, enabling faster regulatory approvals, enhanced patient safety, and global market expansion for specialized analytical labs.
E&L Testing Services Market Segmentation: Parenteral Products and Container-Closure Systems Drive Global Growth
Extractables and Leachables (E&L) Testing Services Market is primarily dominated by high-risk parenteral products, including small-volume parenterals (SVPs) and large-volume parenterals (LVPs), due to their direct bloodstream administration and potential chemical migration risks. Among container-closure systems, vials, stoppers, and seals lead demand as the primary source of extractables and leachables. Growing adoption of advanced analytical testing techniques, regulatory compliance protocols, and risk mitigation strategies positions these segments as key revenue drivers fueling global E&L market expansion.
By Product Tested
Parenteral
Ophthalmic
Topical
Nasal
Others
By Types of container closures and devices tested
Container closure components
Single-use systems
Medical devices and combination products
Electronic nicotine delivery systems
Other
Get Full PDF Sample Copy of Report: (Including Full TOC, List of Tables & Figures, Chart) @ https://www.maximizemarketresearch.com/request-sample/82518/
Regional Insights: North America and Europe Lead Global E&L Testing Services Market with Cutting-Edge Compliance and Biologics Growth
North America dominates the global Extractables and Leachables (E&L) Testing Services Market, driven by FDA-regulated parenteral products, biologics, high-potency injectable drug production, and complex container-closure systems. Advanced container-closure system analysis, state-of-the-art LC-MS/MS, GC-MS laboratories, and multi-modal E&L testing by global CROs accelerate adoption. Rising regulatory compliance demand, toxicological risk mitigation, and innovative analytical workflows position North America as the critical hub for global E&L market growth and patient safety assurance.
Europe ranks as the second-largest region in the Extractables and Leachables (E&L) Testing Services Market, fueled by stringent EMA regulations, high-risk biologics, and high-potency injectable drug manufacturing. Advanced container-closure system evaluation, LC-MS/MS and GC-MS testing platforms, multi-modal E&L services, and specialized CRO networks accelerate regulatory approvals. Europe’s focus on innovative, high-risk pharmaceutical testing solutions and patient safety compliance positions it as a key growth hub in the global E&L testing market.
2025 E&L Testing Market: Eurofins, Intertek, SGS & WuXi Drive Innovation and Compliance
Eurofins Scientific expanded its E&L testing footprint in May 2025 by acquiring specialized testing operations of TestAmerica, strengthening North American analytical services and advanced packaging materials analysis.
Intertek Group plc launched expert Extractables and Leachables (E&L) testing services in October 2025, tailored to new Korean Pharmacopeia mandates to ensure regulatory compliance in pharmaceutical packaging and drug delivery systems.
SGS Societe Generale de Surveillance SA is revolutionizing E&L analysis with state‑of‑the‑art automated sample preparation technologies in 2025, boosting precision, throughput, and regulatory‑ready data integrity.
WuXi AppTec indirectly impacted the E&L testing landscape through industry consolidation as NAMSA’s January 2025 acquisition of WuXi AppTec’s U.S. medical device testing labs broadened biocompatibility and trace E&L service capabilities.
Competitive Landscape:
Extractables and Leachables (E&L) Testing Services Market is intensely competitive, with global leaders like Eurofins Scientific, Intertek Group plc, SGS Société Générale de Surveillance SA, and Merck KGaA driving cutting-edge analytical platforms, advanced LC-MS/MS and GC-MS workflows, and stringent regulatory compliance solutions. Specialist laboratories such as Boston Analytical, Pacific Biolabs, and Sotera Health accelerate multi-modal E&L testing services, while West Pharmaceutical Services and Wickham Micro lead container-closure system analysis and biologics safety testing, positioning this market at the forefront of pharmaceutical quality assurance, patient safety, and global E&L testing innovation and growth.
Extractables and Leachables (E&L) Testing Services Market, Key Players:
Eurofins Scientific
Intertek Group plc
SGS Societe Generale de Surveillance SA
WuXi AppTec
Sotera Health (Nelson Laboratories, LLC)
West Pharmaceutical Services, Inc.
Merck KGaA / Merck & Co.
Smithers (formerly Smithers Rapra)
Labcorp (formerly Covance)
Thermo Fisher Scientific
Pacific Biolabs
Boston Analytical (BA Sciences)
Vimta Labs
Wickham Micro Limited (Medical Engineering Technologies Ltd.)
Agilent Technologies
Element Materials Technology
Sartorius AG
LGC Limited
Charles River Laboratories International
EKG Life Science Solutions (EKG Labs)
Toxikon Corporation
PPD (part of Thermo Fisher)
Alcami
Jordi Labs
VR Analytical
Get access to the full description of the report @ https://www.maximizemarketresearch.com/market-report/global-extractables-and-leachables-testing-services-market/82518/
FAQs:
1: What factors are driving the rapid growth of the Extractables and Leachables (E&L) Testing Services Market globally?
Ans: The E&L Testing Services Market growth is driven by FDA, EMA, and ICH regulations, rising demand for SVP and LVP drug safety, and advanced LC-MS/MS and GC-MS analytical testing, enhancing patient safety and global regulatory compliance.
2: Which segments dominate the Extractables and Leachables (E&L) Testing Services Market, and why?
Ans: High-risk parenteral products (SVPs, LVPs) and container-closure components dominate due to direct bloodstream administration and chemical migration risks, making them critical for regulatory compliance and patient safety.
3: Who are the key players shaping innovation in the global E&L Testing Services Market?
Ans: Leaders like Eurofins Scientific, Intertek, SGS, WuXi AppTec, and Merck KGaA drive multi-modal testing, container-closure analysis, and biologics safety, while specialist labs enhance regulatory compliance and global E&L innovation.
Analyst Perspective:
Extractables and Leachables (E&L) Testing Services sector is evolving rapidly, driven by advanced analytical upgrades, strategic collaborations, and AI integration. Competitive players are expanding regionally, adopting multi-modal testing solutions, and targeting high-risk parenteral products. Robust investment, innovative workflows, and regulatory compliance focus position the market for sustained growth, strategic expansion, and enhanced global adoption.
Related Reports:
Single-use Bioprocessing Market: https://www.maximizemarketresearch.com/market-report/global-single-use-bioprocessing-market/105384/
Bioprocess Validation Market: https://www.maximizemarketresearch.com/market-report/global-bioprocess-validation-market/33430/
Top Reports:
Milk Protein Market
Global Anthocyanin Market
About Maximize Market Research – E&L Testing Services Sector:
Maximize Market Research provides in-depth insights into the Extractables and Leachables (E&L) Testing Services sector, helping clients navigate the complex Material & Chemical domain. Our research identifies regulatory trends, analytical innovations, and risk mitigation strategies, empowering pharmaceutical and biotech companies to optimize container-closure system analysis and ensure global compliance.
Our Expertise in Material & Chemical Analysis:
With a specialized focus on Material & Chemical testing, Maximize Market Research evaluates parenteral, biologics, and high-risk drug products. We guide clients on advanced analytical platforms, multi-modal E&L testing, and container-closure system evaluation, supporting strategic investments, laboratory upgrades, and regional adoption for improved patient safety and regulatory adherence.
Lumawant Godage
MAXIMIZE MARKET RESEARCH PVT. LTD.
+ +91 96073 65656
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.